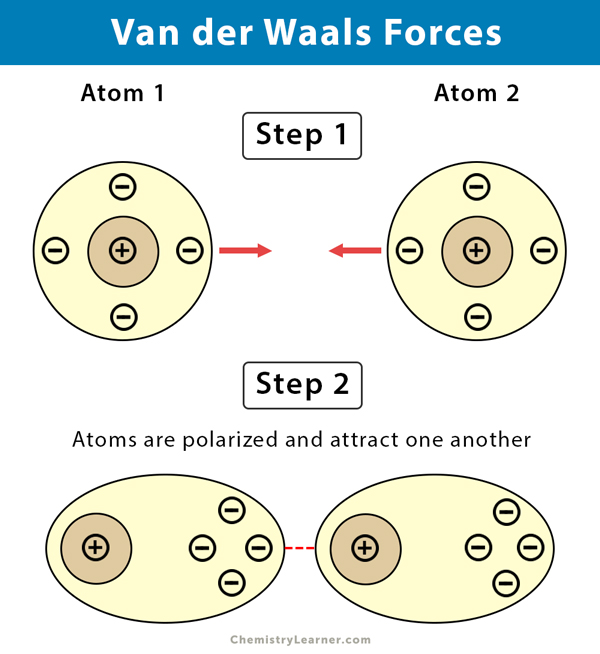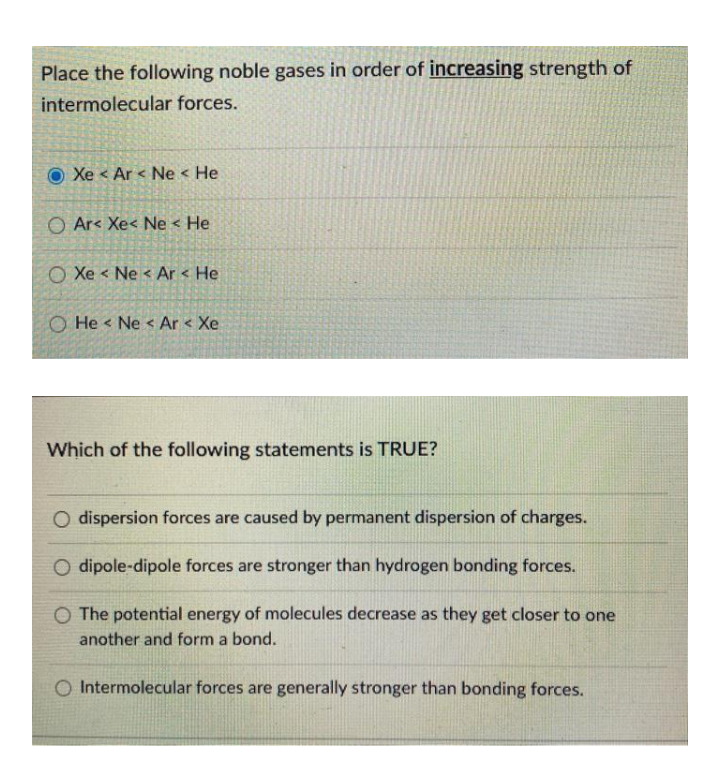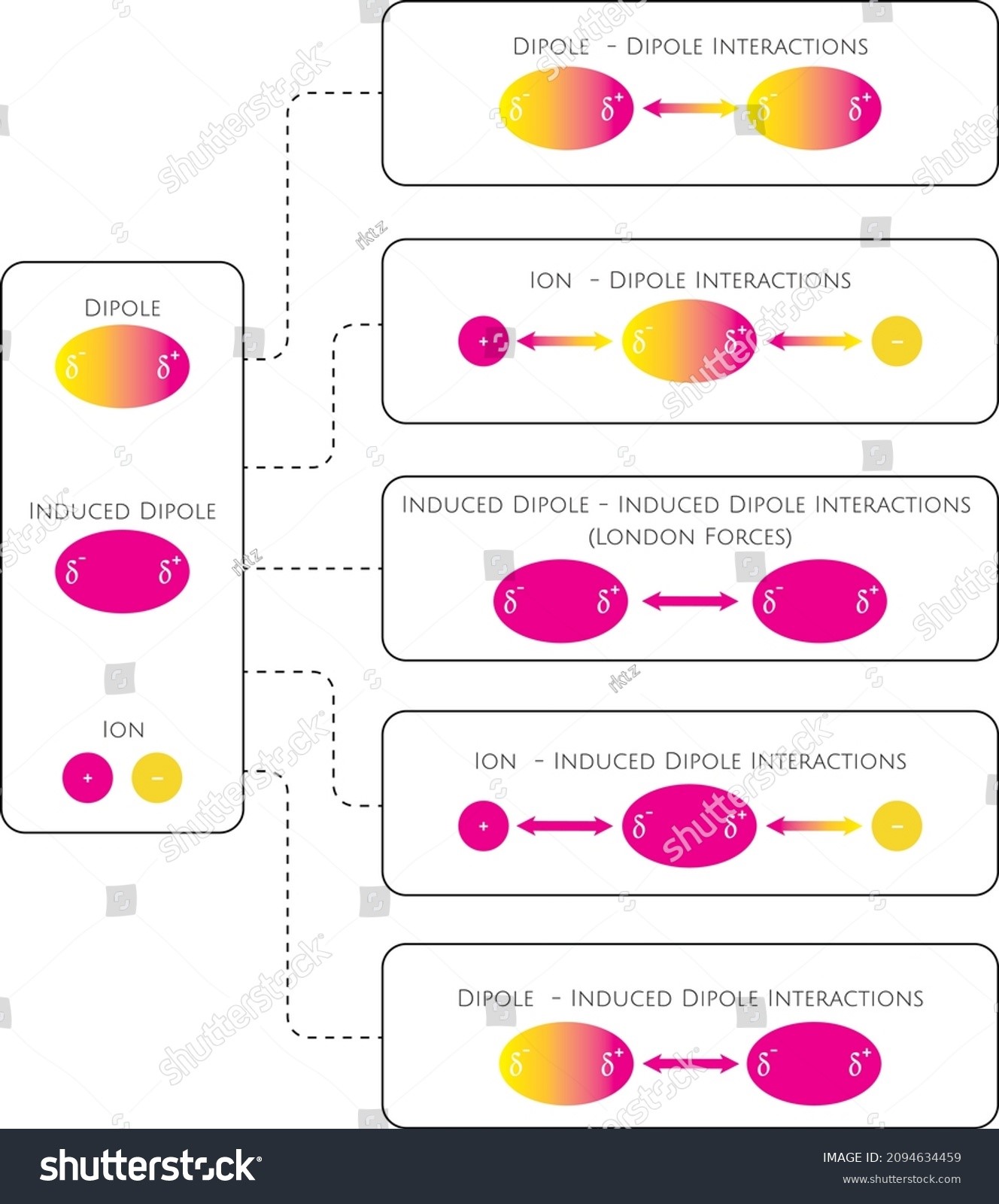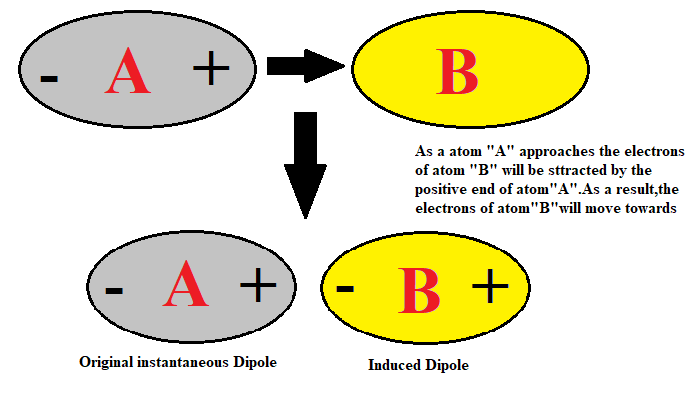
Van der Waals' forces are applied to: A) Inert gases only B) Rate gases only C) Mixture of gasesD) Elementary gases only
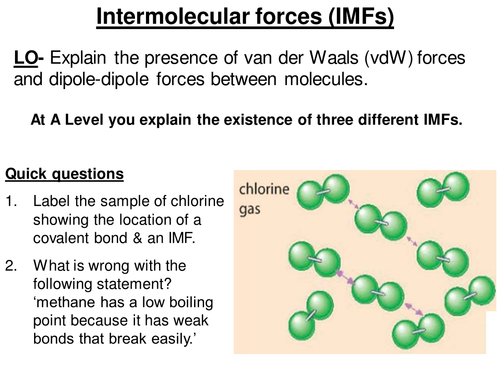
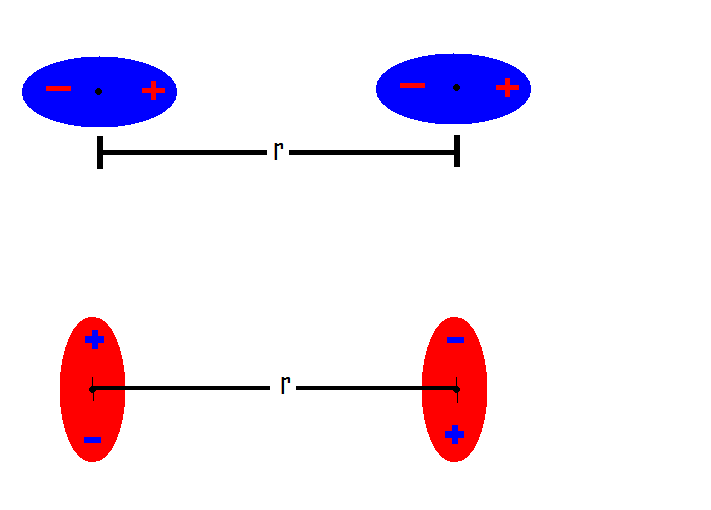
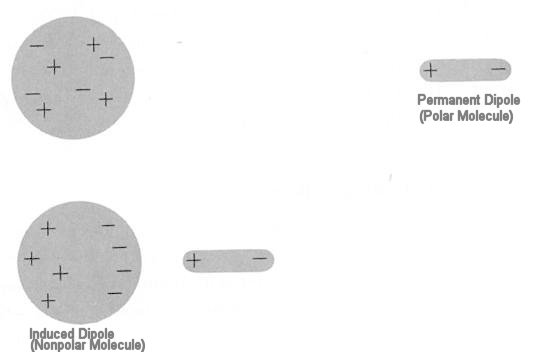
INTERMOLECULAR FORCES/INTERACTIONS Name: Bonding covered so far involved intramolecular bonding or forces, i.e.
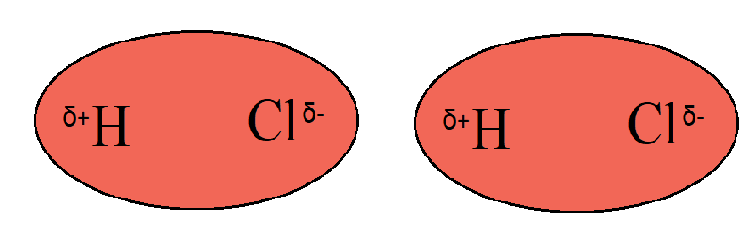
Van der Waals' forces are applied to: A) Inert gases only B) Rate gases only C) Mixture of gasesD) Elementary gases only