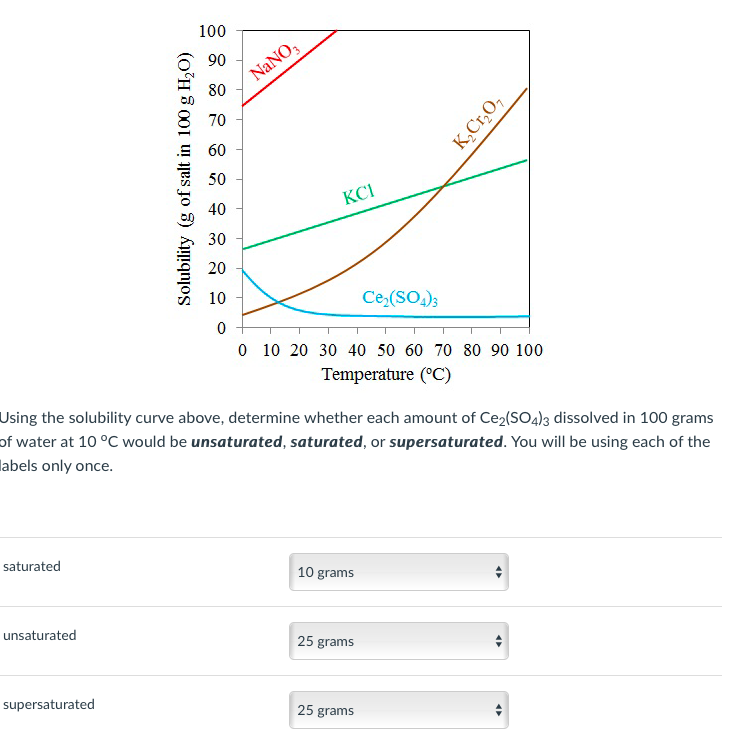
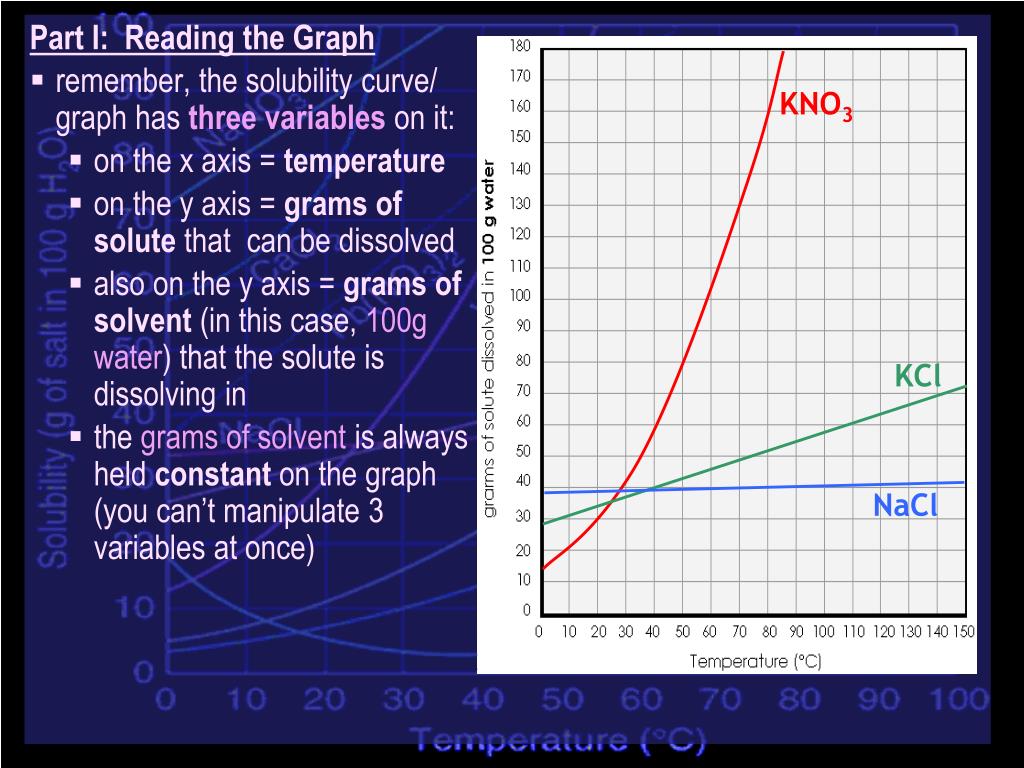
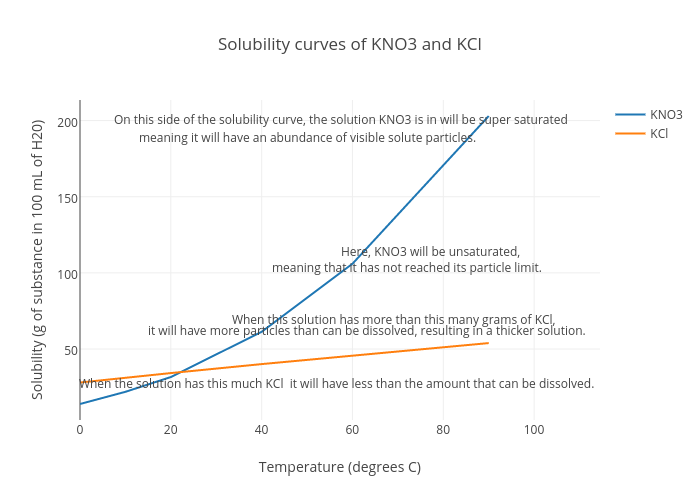
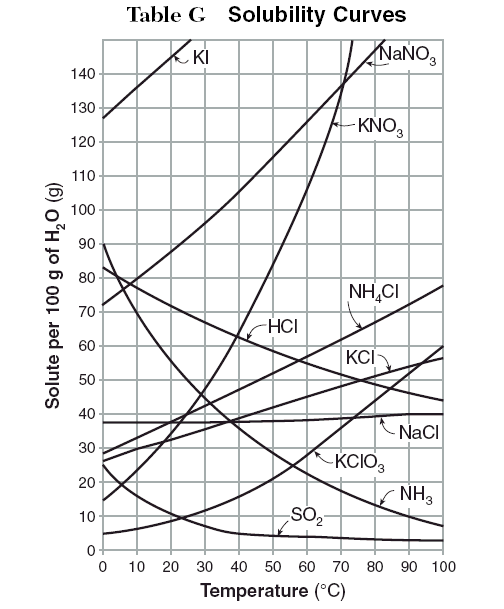
Figure 1 from Growth and characterization of bis ( thiourea ) potassium chloride crystals for NLO applications | Semantic Scholar

Solubility and solvation thermodynamics of dl-nor-valine in aqueous solutions of NaCl and KCl - ScienceDirect
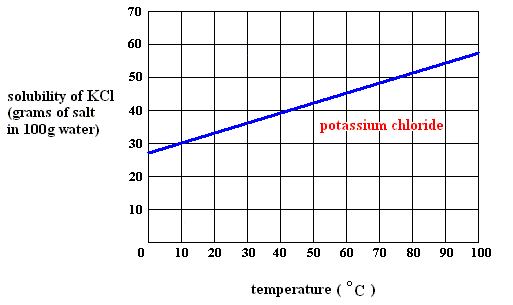
At what temperature is the concentration of a saturated solution of KCl (molar mass 74.5 g) approximately 3 molal? | Socratic
Data Collection: Experiment 4a – Measure the Solubility of Potassium Chloride Experiment 4b – Measure the Solubility of Ammo
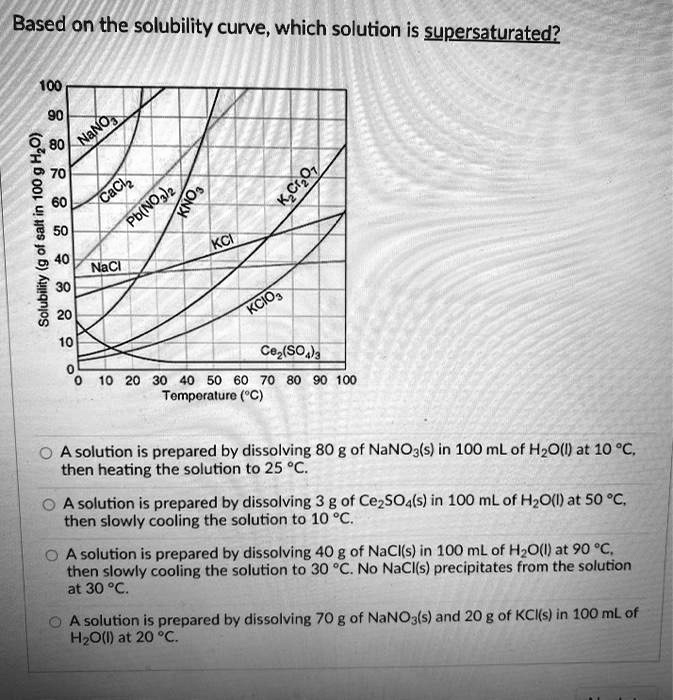
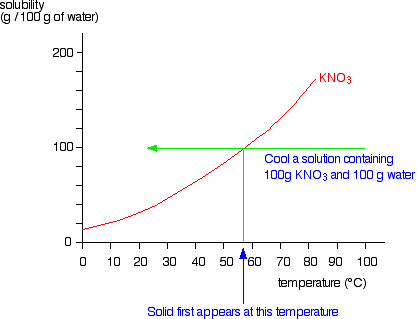
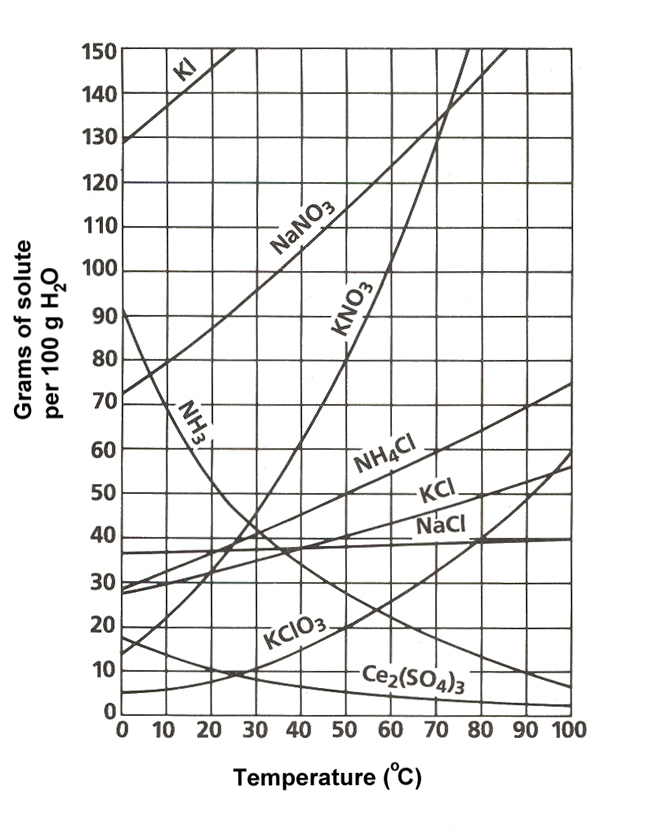
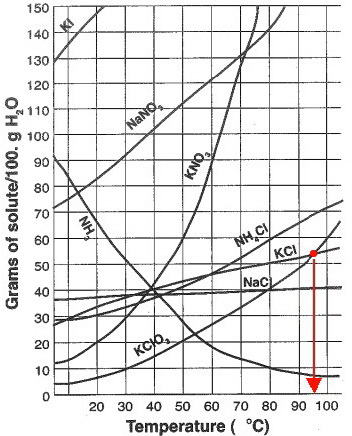
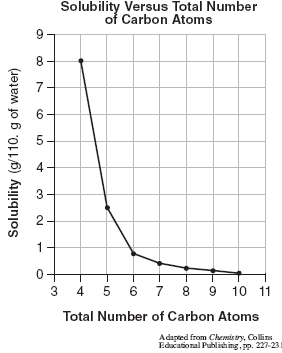
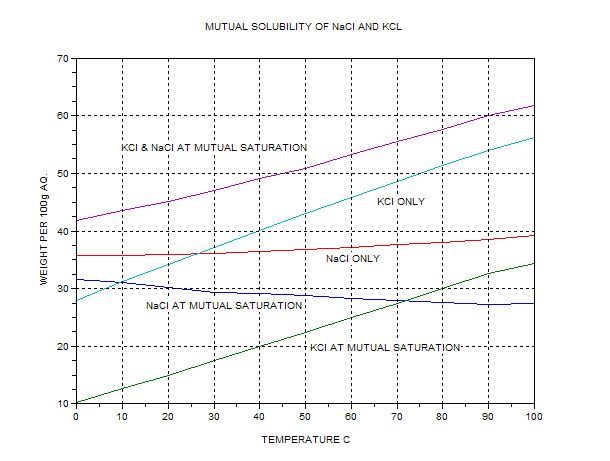
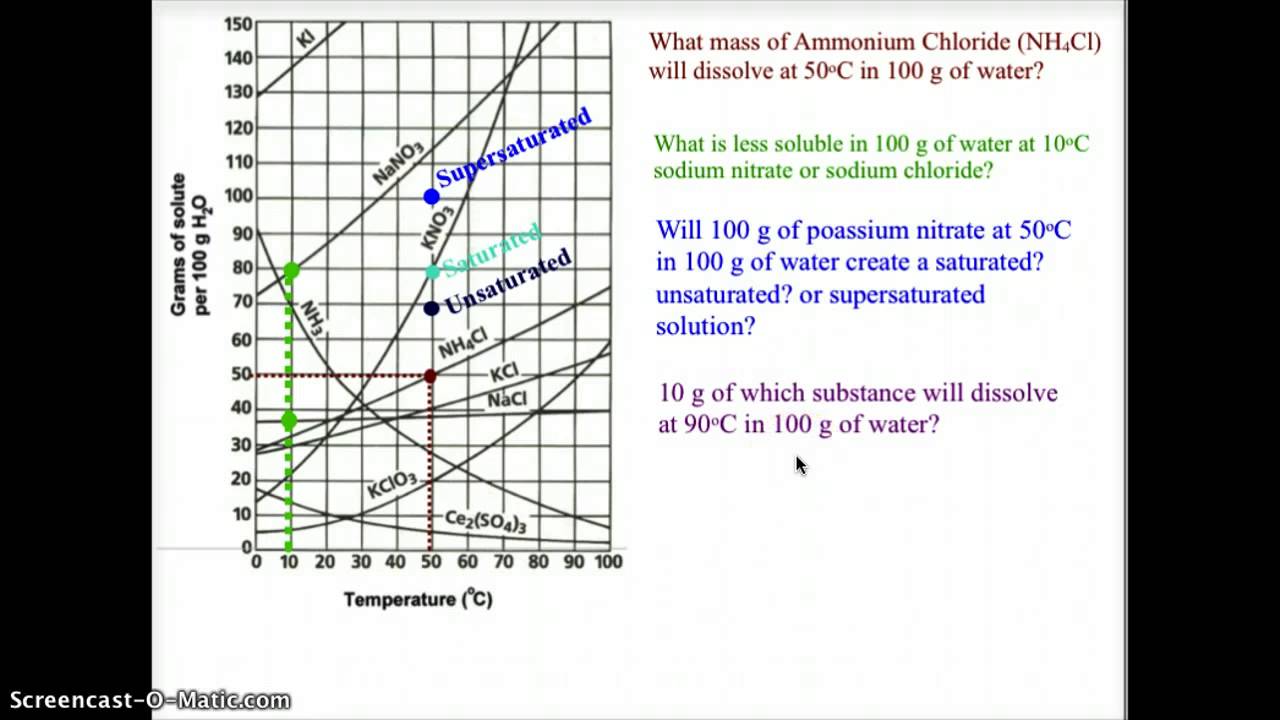
![Ms. Kilroy's Science Classes [licensed for non-commercial use only] / Unit 4 (1) Ms. Kilroy's Science Classes [licensed for non-commercial use only] / Unit 4 (1)](http://mkilroy.pbworks.com/f/Unit+4+%281%29-image-2.gif)