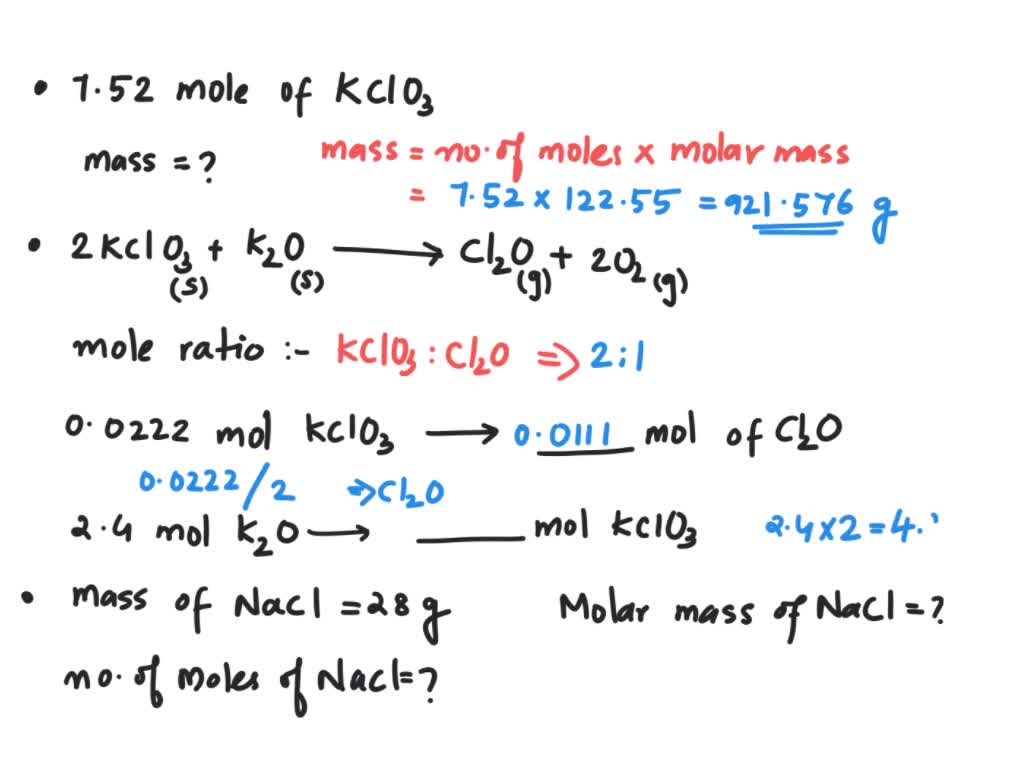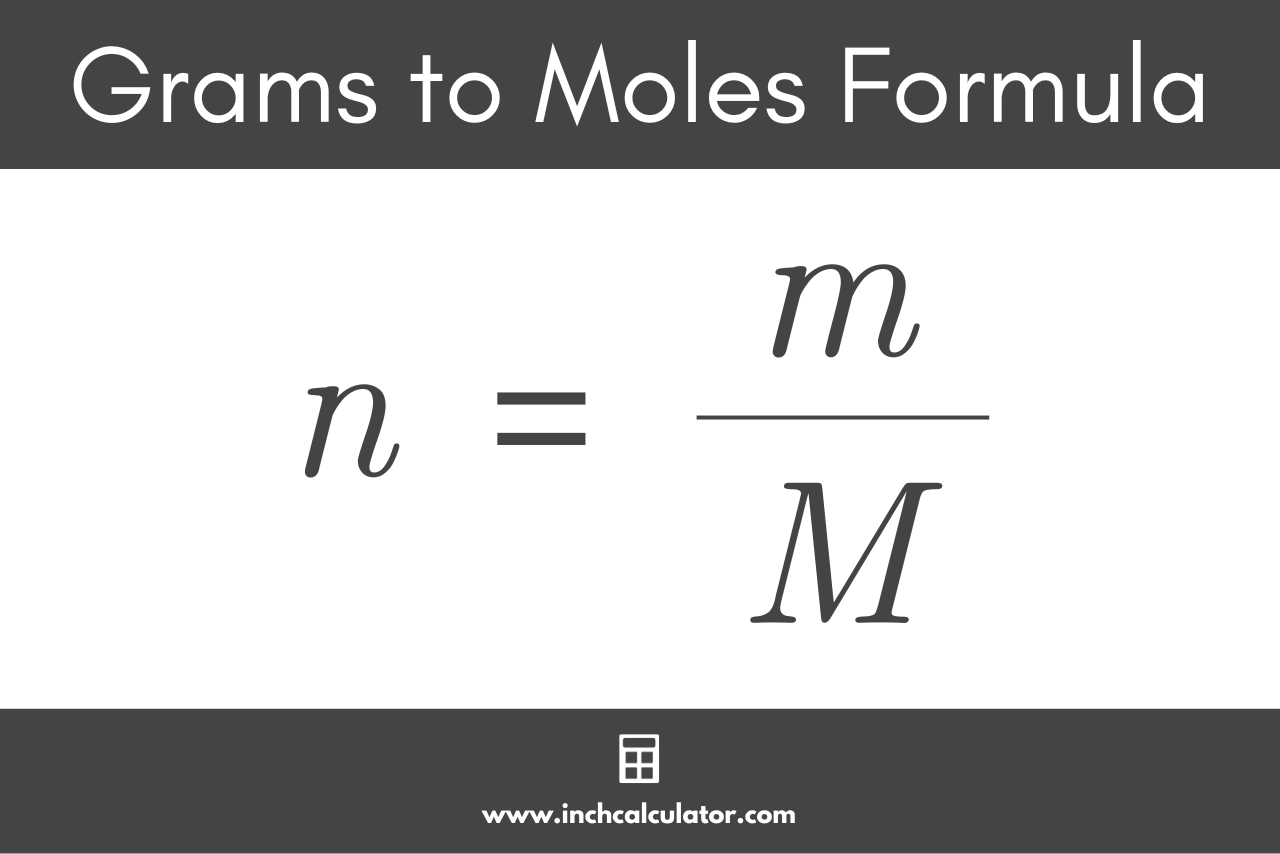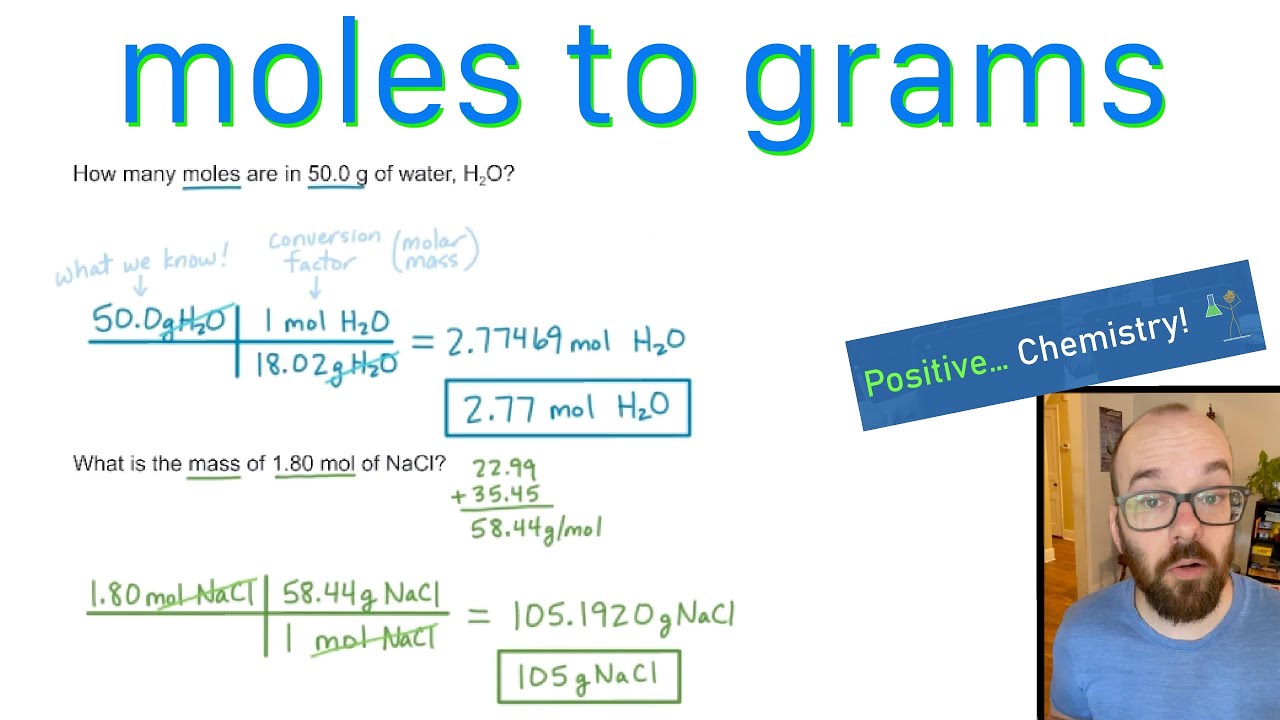Question Video: Determining the Mass of Sodium That Contains the Same Number of Moles as a Given Number of Moles of Copper | Nagwa

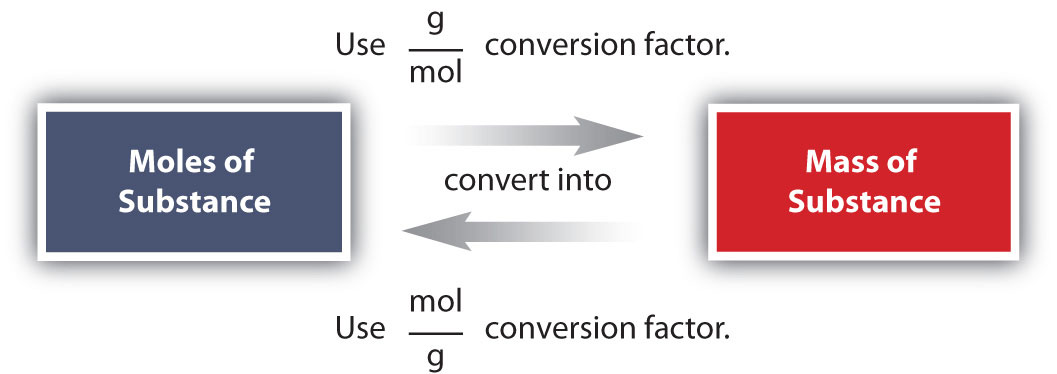
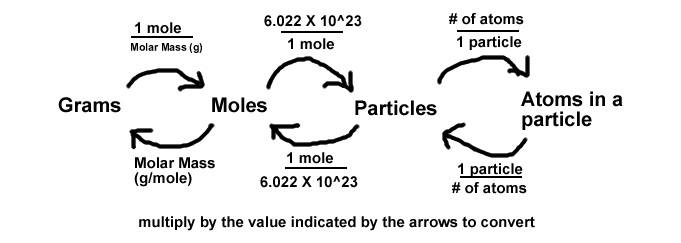
Chemistry Warm Up: Mole / Mass / Particles 1.What is the mass of one mole of water? 2.If one milliliter of water has a mass of 1.00grams, how many moles. - ppt download