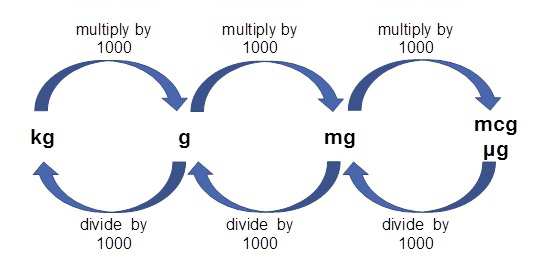
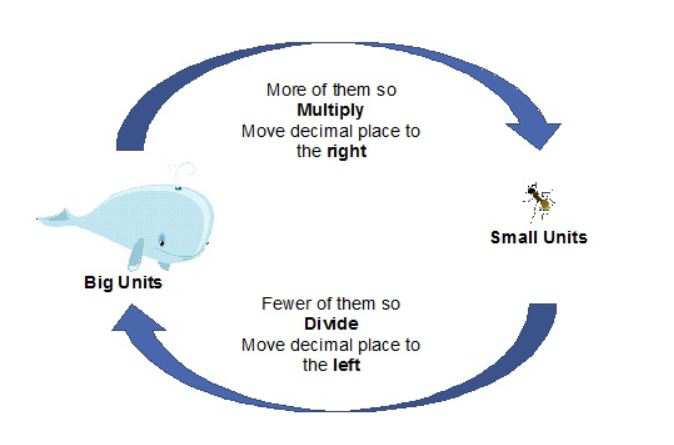
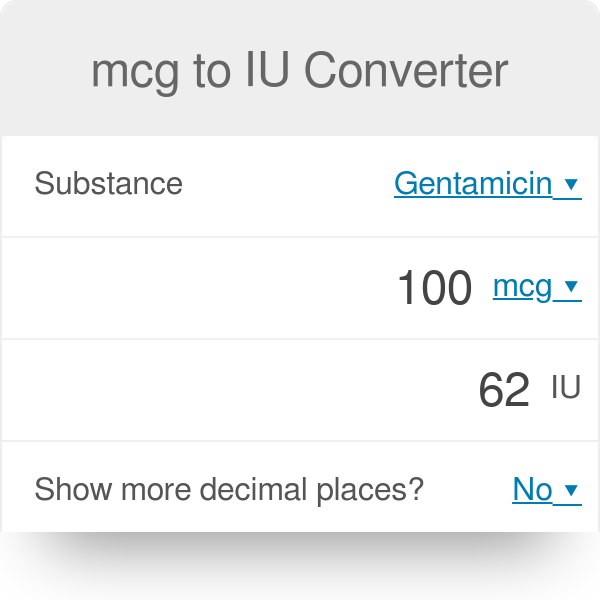
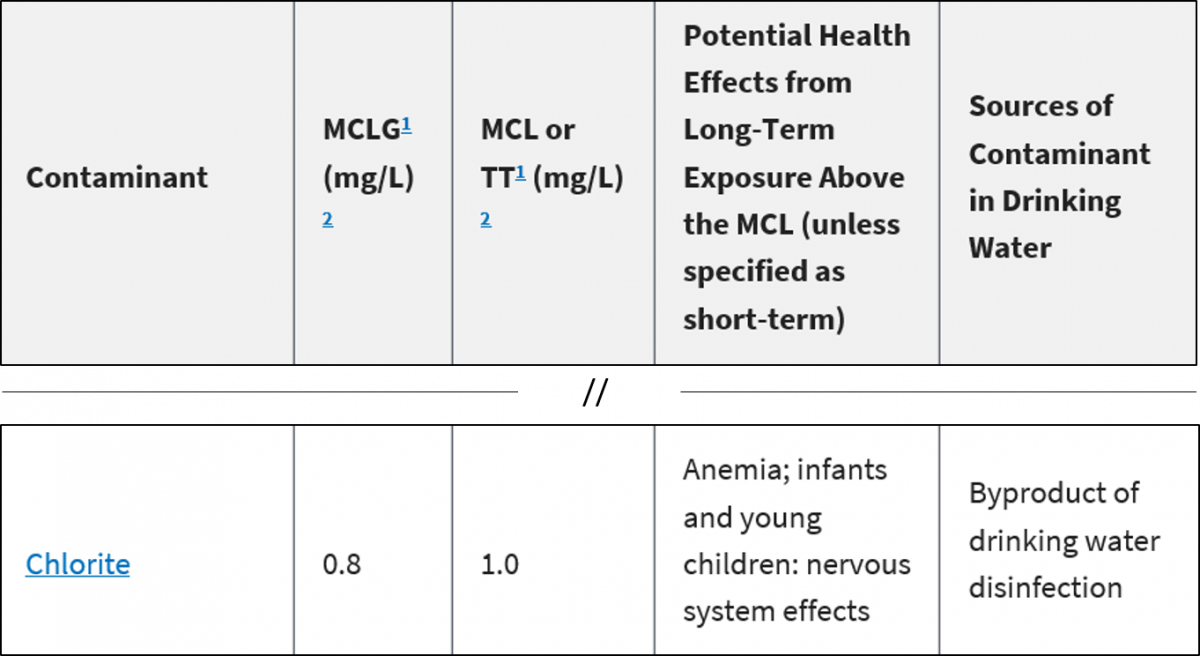
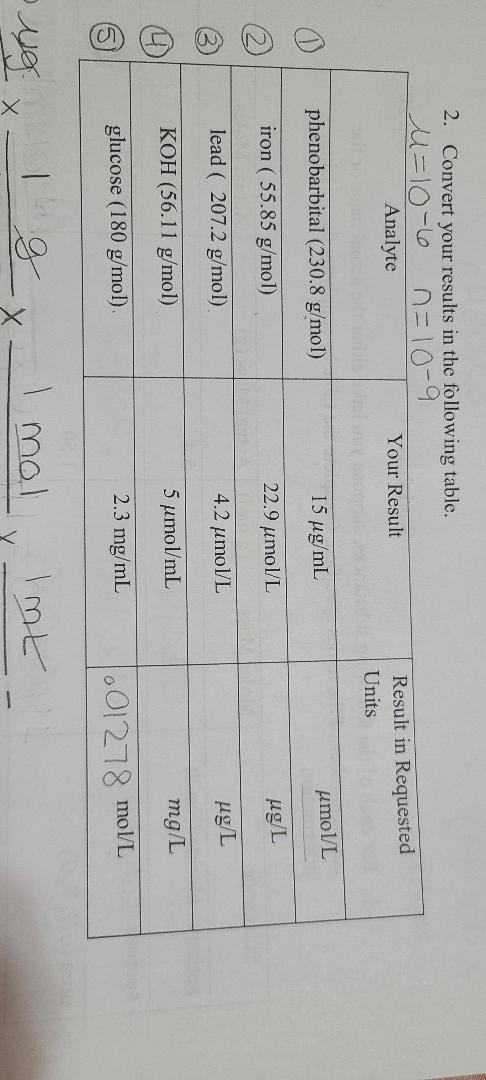
SOLVED: Convert the following (Show your work) (1 point): 98.36 pg/ml 7 ng/ul 347.8 umol 25 ng 9#g 3.2 AM pmol/ul (recall: 1 M = 1 mol/L) pmol

How to convert arsenic concentration ug/L (in solution) to mg/kg (in soil) after sequential extraction experiment ? | ResearchGate






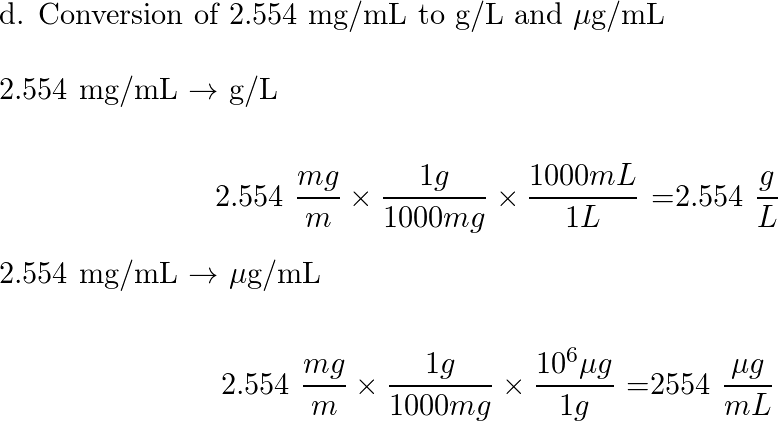


![PDF] Heavy Metal Poisoning and its Laboratory Investigation | Semantic Scholar PDF] Heavy Metal Poisoning and its Laboratory Investigation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7db8bfa7e9d6ce9010dba9da905b1fdf245207ec/34-Table13-1.png)