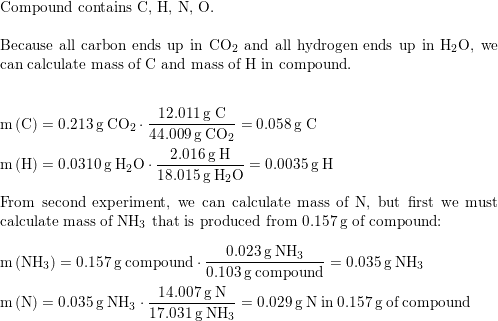Butyric acid contains only C,H and O.a4.24 mg sample of butyric acid is completely burned. It gives 8.45 mg of CO2 and 3.46 mg of H2O .The molecular mass of butyric acid

Acetic acid contains only C, H, and O. A 4.24-mg sample of acetic acid is completely burned. It gives 6.21 - Brainly.com
✓ Solved: You perform a combustion analysis on a 255 mg sample of a substance that contains only C, H,...
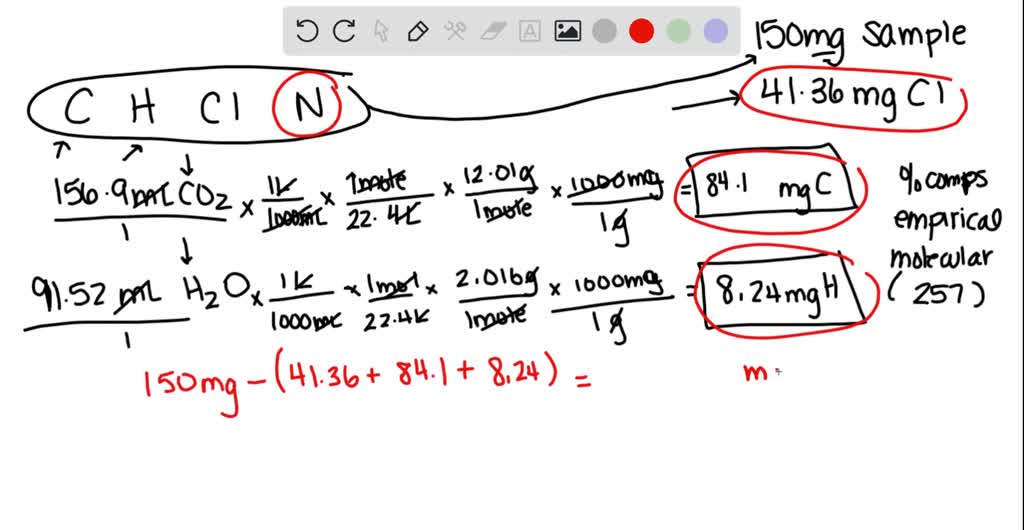
SOLVED: An herbicide contains only C, H, Cl, and N. The complete combustion of a 150.0 mg sample of the herbicide in excess oxygen produced 156.9 mL of CO2 and 91.52 mL
How can I determine the molecular formula for a compound that contains only C and H and has a molecular ion with an m/z value of 72? | Socratic

Draw the structure of an alkane with molecular formula C7H16 that contains (a) one 4 carbon; (b) only 1 and 2 carbons; (c) 1 , 2 , and 3 hydrogens. | Homework.Study.com

A compund contains only C,H and N Combustion of 70mg of the compound produces 132mg CO2 and 54mg H2O - Chemistry - Some Basic Concepts of Chemistry - 13742173 | Meritnation.com
What is the logic for whether the string is complete (contains all letters from a to z) in C? - Quora

SOLVED: 'Question 11 of 13 Psoralen, a compound found in giant hogweed, contains only C, H, and O. Combustion of 1.000 g of psoralen produces 2.600 g of CO and 0.2903 g

SOLVED: Xanthotoxin, a photosensitizer compound found in parsnips, contains only C, H, and O. Combustion of 1.000 g of xanthotoxin produces 2.443 g of CO? and 0.3334 g of H?O. Determine the
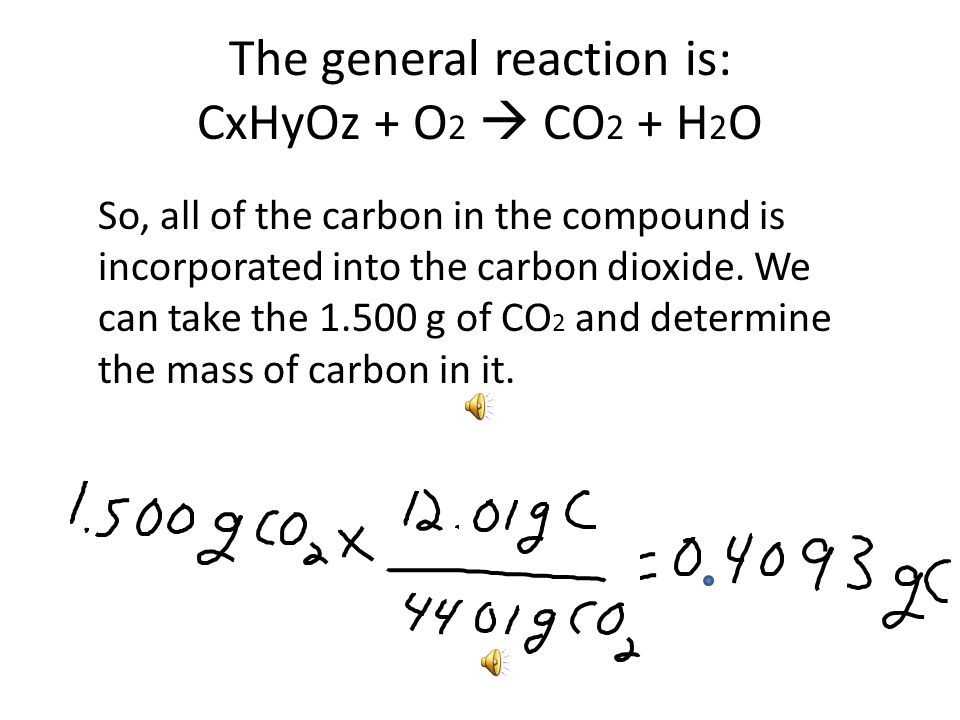
Combustion Analysis Ex - Find the empirical formula of vitamin C (ascorbic acid), a compound that contains only C, H, and O. Combustion of g of. - ppt download

sample of pure boron contains only isotope X and isotope Y. A nucleus of X has more mass than a nucleus of - Brainly.com
A 4.24 mg sample of butyric acid is completely burnt.It gives 8.45 mg of carbon dioxide and 3.46 mg of H 2 O what is mass percent of each element in butyric